Purity and separating mixtures
C2: Elements, compounds and mixtures
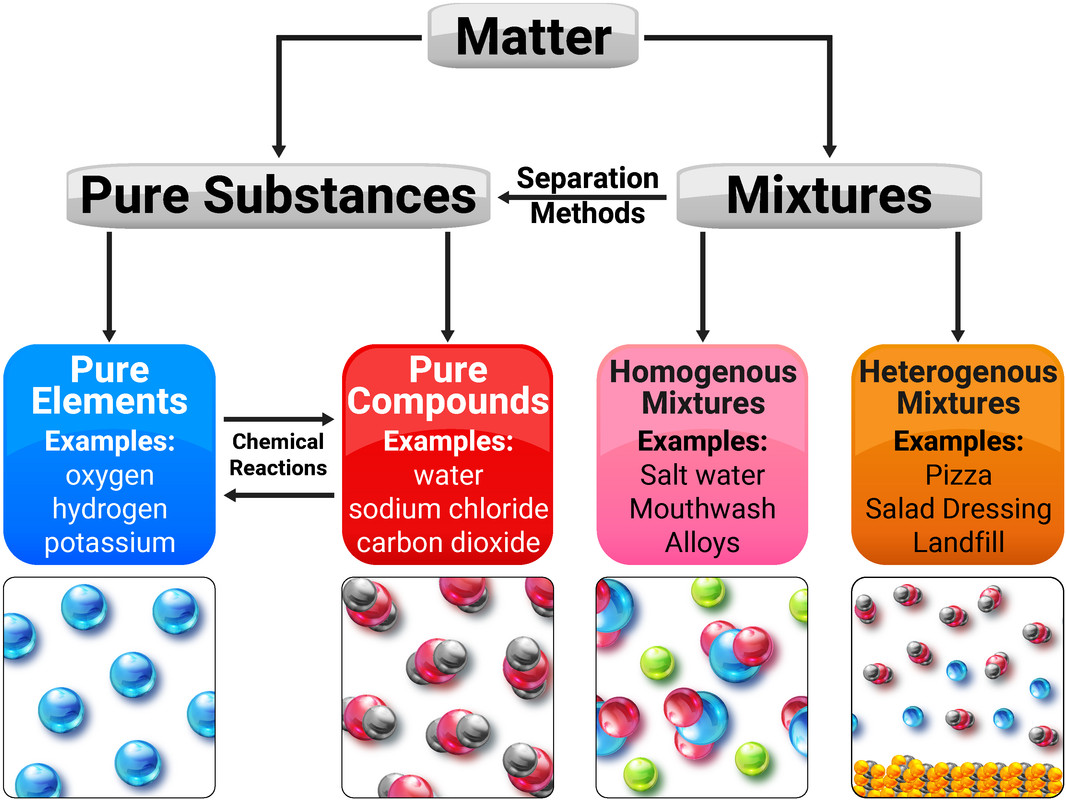
Pure Substances
An element contains atoms with the same atomic number.
A compound contains two or more elements chemically joined together.
A mixture consists of two or more elements/compounds, that are not chemically combined together and can be separated by physical processes.
A pure substance is either a single element, or compound, that is not mixed with any other substance.
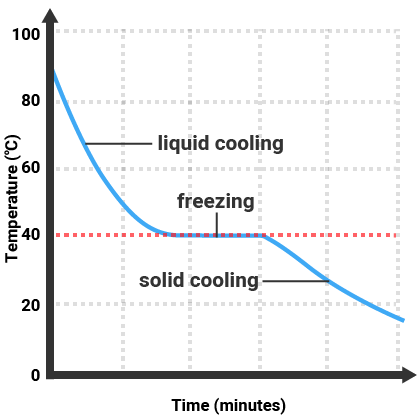
Pure substances will melt and boil at specific temperatures, so we can use the temperature that a substance melts/boils at to tell how pure it is. Impure substances will melt/freeze or boil/condense over a range of temperatures.
 |
 |
| Pure Substance |
Impure Substance |
Pure Substances
An element contains atoms with the same atomic number.
A compound contains two or more elements chemically joined together.
A mixture consists of two or more elements/compounds, that are not chemically combined together and can be separated by physical processes.
A pure substance is either a single element, or compound, that is not mixed with any other substance.
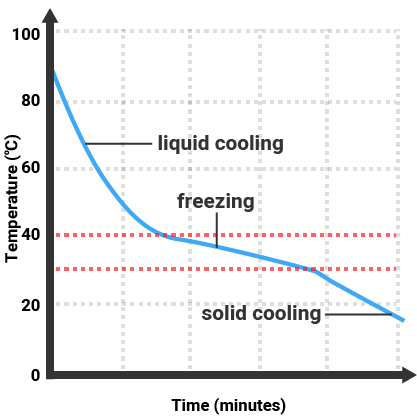
Pure substances will melt and boil at specific temperatures, so we can use the temperature that a substance melts/boils at to tell how pure it is. Impure substances will melt/freeze or boil/condense over a range of temperatures.
 |
 |
| Pure Substance |
Impure Substance |

Formulations
A formulation is a mixture that has been designed as a useful product. Many products are complex mixtures in which each chemical has a particular purpose. Formulations are made by mixing the components in carefully measured quantities to ensure that the product has the required properties. Formulations include fuels, cleaning agents, paints, medicines, alloys, fertilisers and foods.
It is important that formulations keep the same ingredients, so that the formulation is always produced the same way. People buy these products for continuity, especially paints - it would be pointless buying the same shade of paint that was actually different colours.

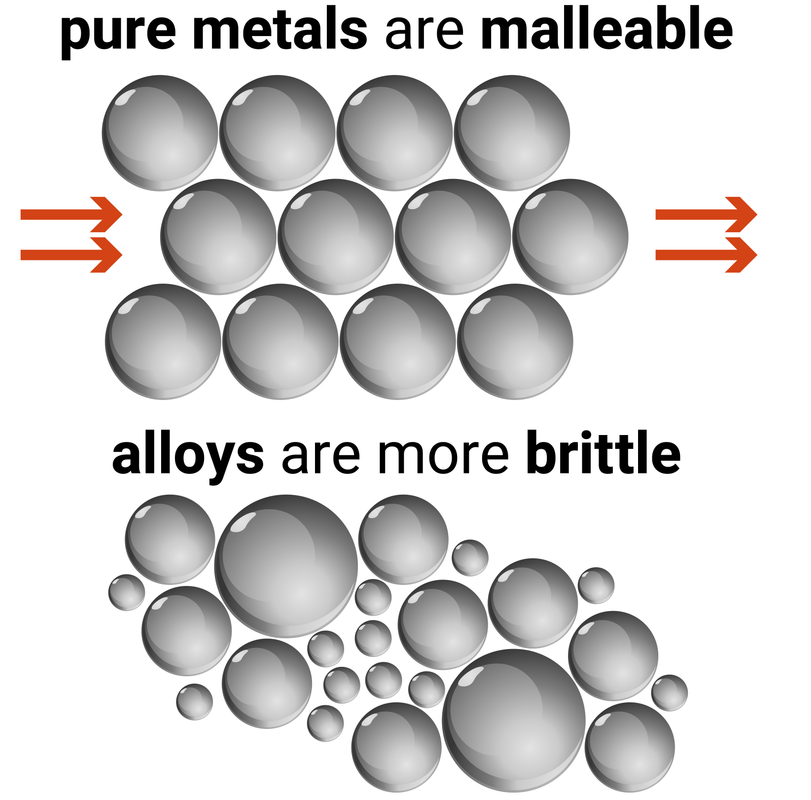
Most metals in everyday use are alloys. An alloy is a mixture of two or more elements, where at least one of the elements is a metal.
Pure metals are generally too soft to use as they have a regular lattice structure. When a force is applied to the metal, the regularly aligned layers are able to easily slide over each other (this makes them malleable).
Alloys are made of different sized atoms, distorting the regular layers so that they are not able to easily slide over each other any more - increasing the strength of the metal (they are less malleable, and more brittle).
Separation Techniques
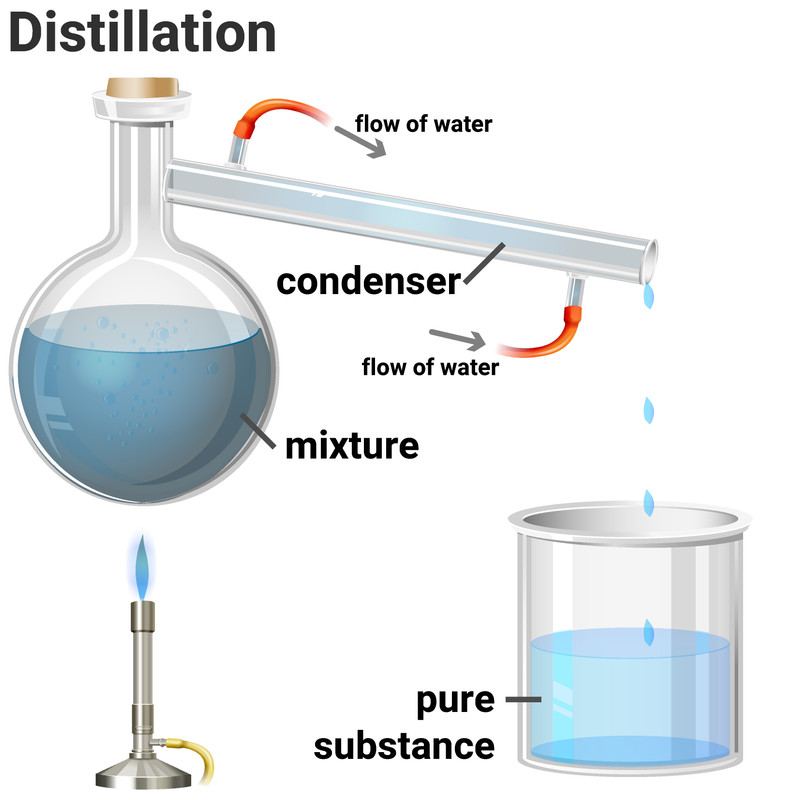
Simple distillation is a method for separating the solvent from a solution, leaving behind the solute. This method works because the solvent has a much lower boiling point than the dissolved solute. Heating the solution allows the solvent to evaporate, and then it passes through a condenser, where it is cooled and condensed into a separate container. The solute does not evaporate and so it stays behind.
Fractional distillation is a method of separating multiple liquids from each other. The technique works in the same way as distillation, but on a much larger scale.
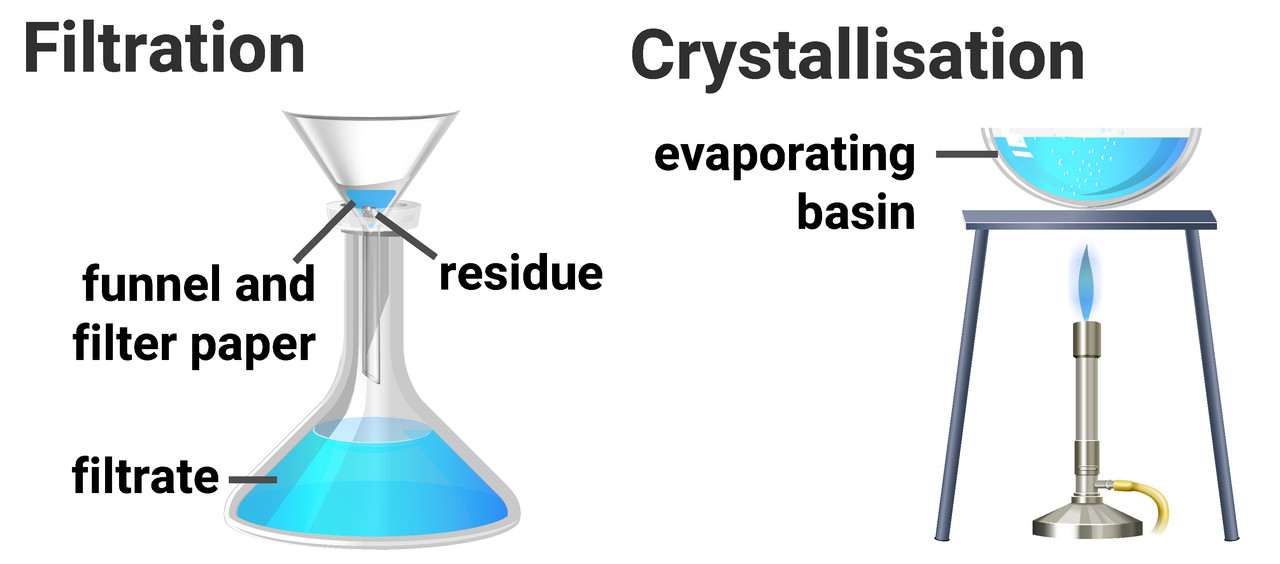
Filtration is a method for separating an insoluble solid from a liquid. A mixture of liquid and solid is passed through filter paper into a flask below. The solid stays on the filter paper (residue), and the liquid passes through to the container below (filtrate).
Crystallisation/evaporation is a method that separates a soluble solid from a liquid. A solution of liquid and dissolved solid is heated, causing the solvent to evaporate and leaves solid crystals behind.
Chromatography
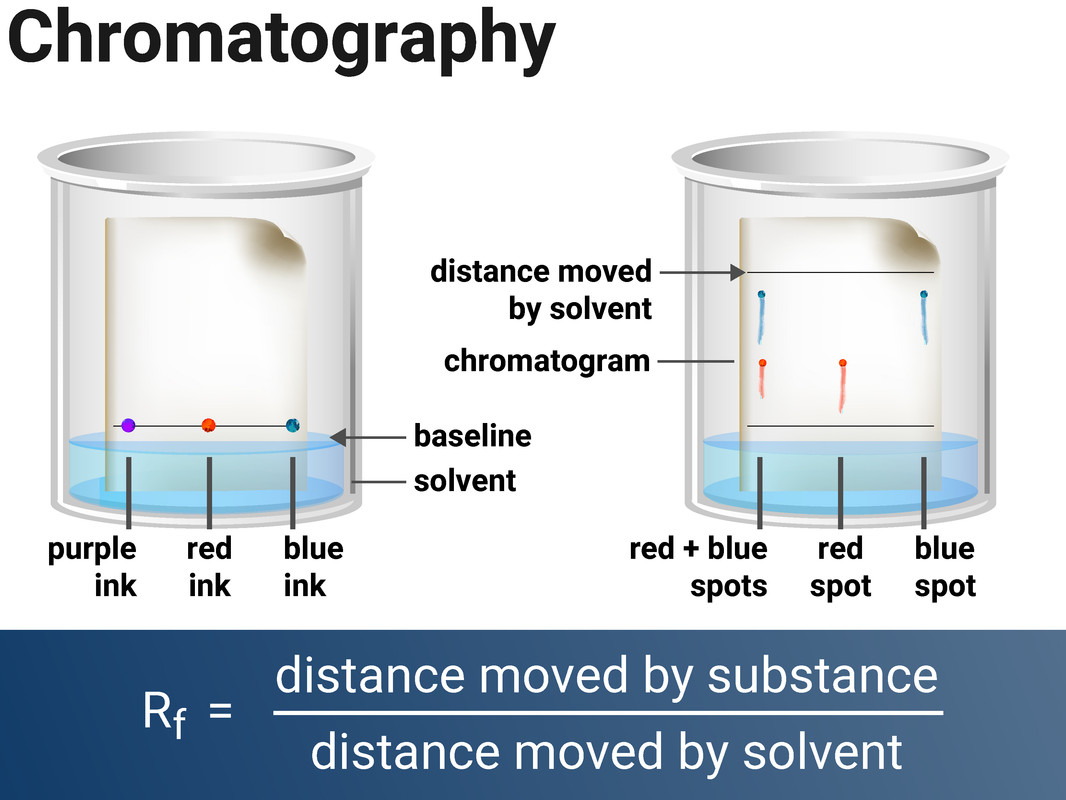
Chromatography is a method for separating dissolved substances from one another. It works because some of the coloured substances dissolve in the solvent better than others, so they travel further up the paper. This technique is often used to separate out inks, or food colourings.
Paper chromatography consists of two stages:
- the stationary phase (a.k.a. the phase that doesn't move - this is the paper)
- the mobile phase (a.k.a. the phase that moves - this is the solvent)
Separation by chromatography produces a chromatogram, and can be used to distinguish between a pure substance (that produces one spot), and a mixture (impure) which produces multiple spots. Different substances will move at different rates through the paper.
When setting up the experiment you must make sure to draw the baseline in pencil, otherwise the ink from a pen would also dissolve in the solvent.
The Rf value of a spot can be used to identify unknown chemicals if they can be compared to a range of reference substances. The Rf value will always be the same for each substance (when the same solvent is used), and an Rf value will always have a value of less than 1. It can be calculated by using:
Rf value = distance moved by substance ÷ distance moved by solvent
Gas chromatography
Gas chromatography (GC) onsists of two stages:
- the mobile phase (an unreactive gas such as nitrogen or helium)
- the stationary phase (very thin layer of an unreactive liquid)
The stationary phase is held on an unreactive solid support (e.g. silica beads). These are packed into a long, coiled ‘column’ inside a temperature-controlled oven. A detector then measures the amount of each substance in a mixture as it leaves the column:
- different substances will travel at different speeds through the column
- these substances will leave at different times, called retention times
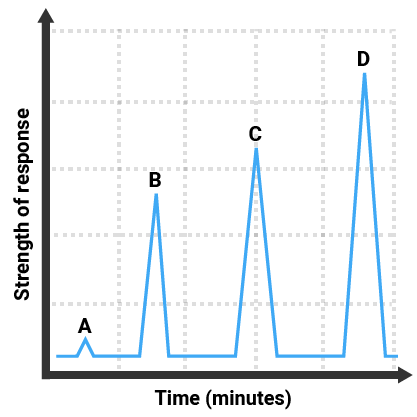
A graph of detector reading against retention time will be plotted, and three main pieces of information can be gathered:
- the number of compounds in the mixture - the number of peaks
- the amount of each compound present - the height of each peak (higher = more)
The gas chromatogram here shows that:
- four different substances were in the mixture (there are four peaks)
- A was present in the smallest amount (it has the smallest peak)
- D was present in the greatest amount (it has the largest peak)