
Reactions of Alkenes and Alcohols (chemistry only)
Organic Chemistry
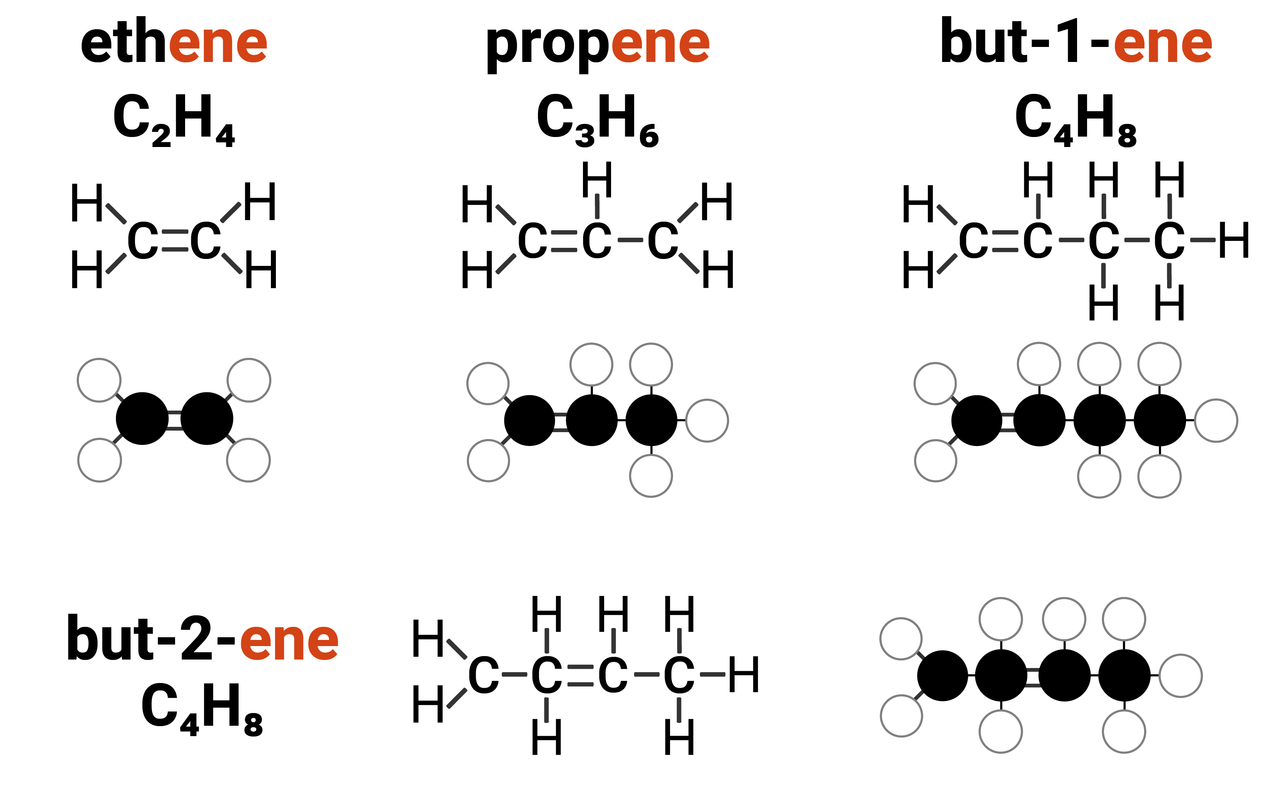
Homologous Series: Alkenes
Alkenes are one example of a homologous series. They:
- have the general formula of CnH2n
-
this means that for every 1 carbon atom in an alkane, there are two times the amount of hydrogens
- differ by CH2 in molecular formulae from neighbouring compounds
-
increasing the carbon chain length by 1 carbon atom, also increases the number of hydrogens by 2
- show a gradual variation in physical properties
-
boiling points increase with increased carbon chain length
viscosity increases with increased carbon chain length
- have similar chemical properties
Because alkenes contain at least one double bond between carbon atoms, we describe them as unsaturated compounds. Think of when a sponge cannot hold any more water, it is "full". A saturated molecule can't fit any more single bonds into it. Alkenes can fit more single bonds in (once the double bonds are broken).
Alkenes can have more than one carbon-carbon double bond, however you will only ever be expected to draw an alkene with one of these bonds.
For ethene and propene, the double bond can only go in one place, however for any chains longer than 3 carbons - this can get a bit more complicated. You need to know the structures of all the isomers of butene that can form due to the double bond being in one of two places.
Reactions of Alkenes
Addition reactions
The functional group (double carbon bond, C=C) allows alkenes to undergo addition reactions. The reaction is an 'addition' reaction because one molecule combines with another molecule, forming one larger molecule and no other products.
The test for alkenes makes use of the addition reaction, and involves adding bromine water to a sample of hydrocarbon. If an alkane is present the solution stays orange-brown, but an alkene will turn the solution colourless. This is because the bromine is added across the carbon-carbon double bond.
Combustion
Alkenes react with oxygen in combustion reactions in the same way as other hydrocarbons, but they tend to burn in air with smoky flames because of incomplete combustion.
The complete combustion of alkanes and alkenes involves the oxidation of both the carbon and hydrogen atoms in hydrocarbons, producing carbon dioxide and water. For example:
ethane + oxygen → carbon dioxide + water
2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l)
ethene + oxygen → carbon dioxide + water
C2H4(g) + 3O2(g) → 2CO2(g) + 2H2O(l)
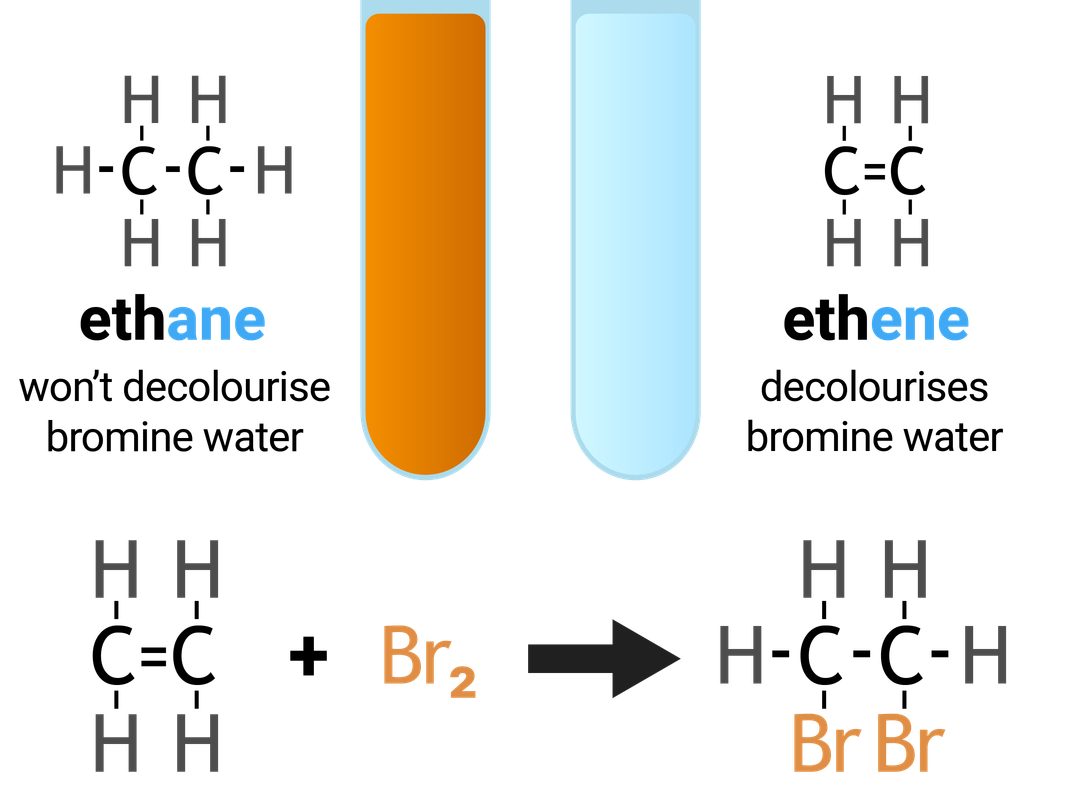
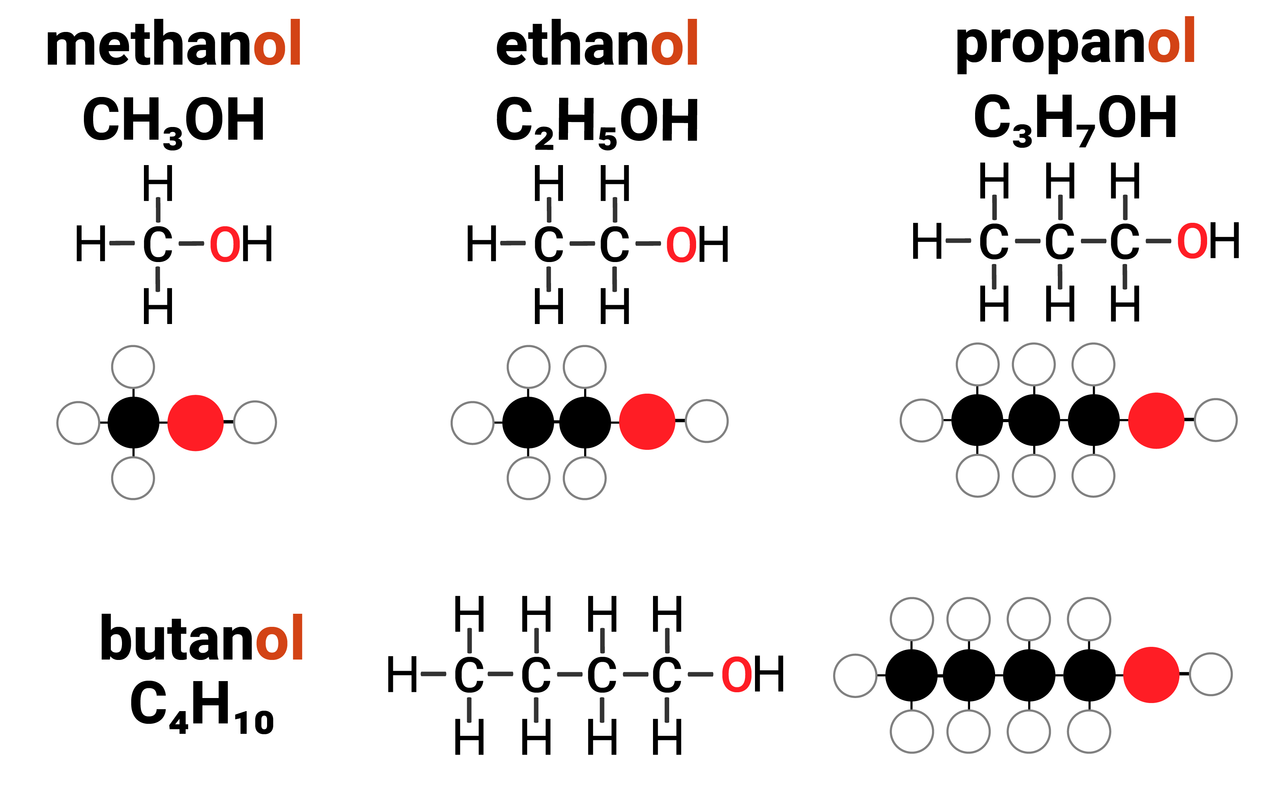
Homologous Series: Alcohols
Alcohols are one example of a homologous series. They:
- have the general formula of CnH2n+1OH
-
this means that for every 1 carbon atom in an alcohol, there are two times the amount of hydrogens... plus one more, and an OH group
- differ by CH2 in molecular formulae from neighbouring compounds
-
increasing the carbon chain length by 1 carbon atom, also increases the number of hydrogens by 2
- show a gradual variation in physical properties
-
boiling points increase with increased carbon chain length
viscosity increases with increased carbon chain length
- have similar chemical properties
Alcohols have the functional group of -OH (hydroxyl) which gives rise to the ending of alcohol. It is responsible for the general reactions that all alcohols can do.
The longer the carbon chain of an alcohol, the more energy the molecule stores. When burned, this energy is released in the form of heat and can be measured using a calorimeter.
Production of Ethanol
Fermentation
glucose → ethanol + carbon dioxide
Most ethanol is made from plants, making it a renewable resource. The process is called fermentation, and requires:
- water
- yeast enzymes
- an absence of oxygen (an anaerobic process)
- temperatures between 25 - 35 °C
The yeast dies when the ethanol concentration reaches about 15%.
Fermentation is a slow reaction and takes several days or weeks to finish. If any air is present, the oxygen oxidises the ethanol to make ethanoic acid, making the drink taste of vinegar.
Purification
Fermentation will produce a dilute solution of ethanol, and needs to be purified to remove water and other liquids (removed by fractional distillation), insoluble substances (removed by filtration), to produce pure ethanol.
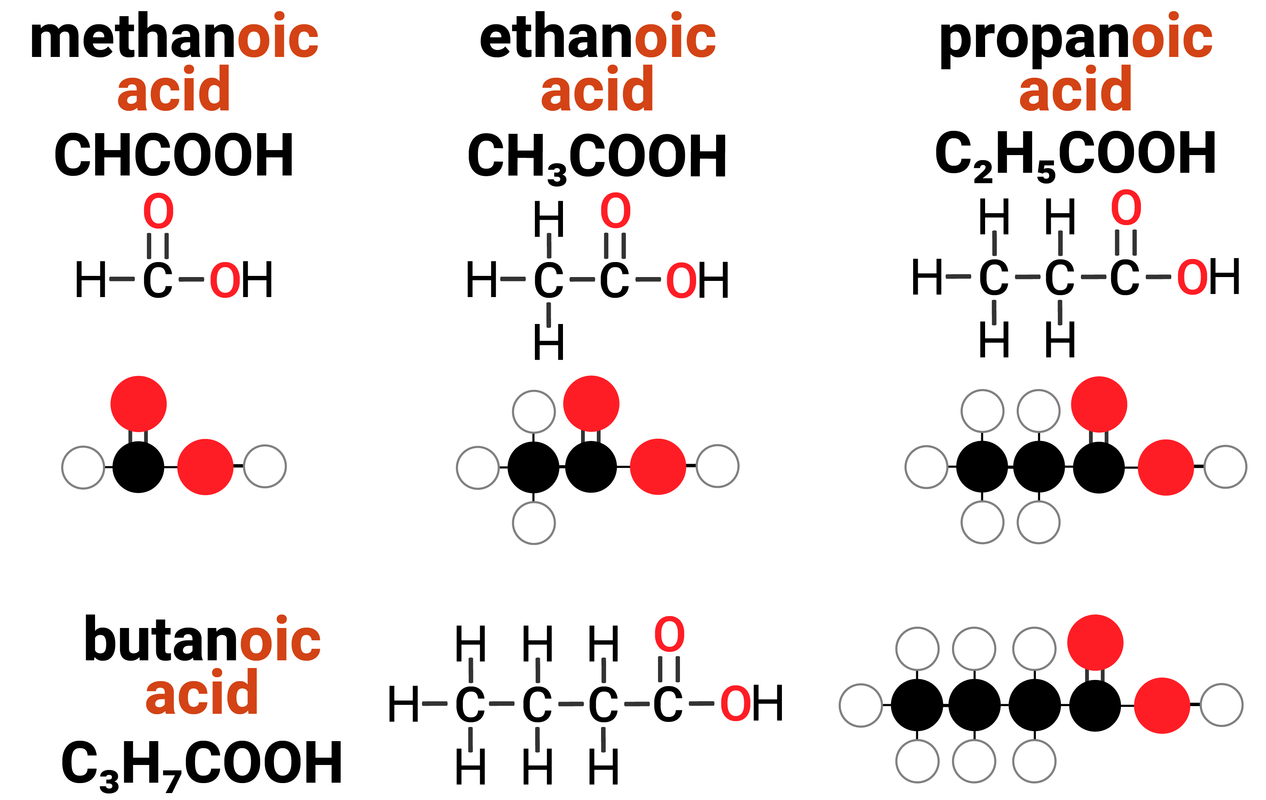
Homologous Series: Carboxylic Acids
Carboxylic acids are one example of a homologous series. They:
- have the general formula of CnH2n+1COOH
-
this means that for every 1 carbon atom in a carboxylic acid, there are two times the amount of hydrogens... plus one more, and a COOH group
- differ by CH2 in molecular formulae from neighbouring compounds
-
increasing the carbon chain length by 1 carbon atom, also increases the number of hydrogens by 2
- show a gradual variation in physical properties
-
boiling points increase with increased carbon chain length
viscosity increases with increased carbon chain length
- have similar chemical properties
Carboxylic acids have the functional group of -COOH (carboxyl) which gives rise to the ending of carboxylic acid. It is responsible for the general reactions that all carboxylic acids can do.
Reactions of Alcohols and Carboxylic Acids
Alcohols
- colourless liquids that dissolve in water to form neutral solutions
- can be dehydrated to form alkenes (produces a water molecule)
- will react with the alkali metals to produce hydrogen and a metal hydroxide (like sodium would with water)
- burn in air to produce carbon dioxide and water (complete combustion)
- can be oxidised to make a carboxylic acid in the presence of an oxidising agent (shown as [O])
Carboxylic acids
- dissolve in water to form acidic solutions (pH values less than 7)
- react with metals to form a salt and hydrogen
- react with bases to form a salt and water
- react with carbonates to form a salt, water and carbon dioxide