Apparatus and techniques that the practical must use or cover
Safe use and careful handling of gases, liquids and solids, including careful mixing of reagents under controlled conditions, using appropriate apparatus to explore chemical changes and/or products
Risk Asessment
As a general rule, eye protection (goggles) must be worn for all practicals.
| hazard | possible harm | precaution |
|---|---|---|
| chlorine solution |
chlorine gas is produced, which is toxic if inhaled |
well ventilated lab; avoid inhaling the gas |
| bromine solution |
bromine gas is produced, which is toxic if inhaled; solution irritates the skin | well ventilated lab; avoid inhaling the gas; wear gloves |
| iodine solution |
harmful to the skin; stains clothes | wear gloves; avoid contact with clothes |
This risk assessment is provided as an example only, and you must perform your own risk assessment before doing this experiment.
Apparatus
Each group will need:
spotting tile
chlorine water
bromine water
iodine water
dropping pipettes
potassium salts (chloride, bromine, chloride)
Experiment Set-up
Simple Distillation
Method
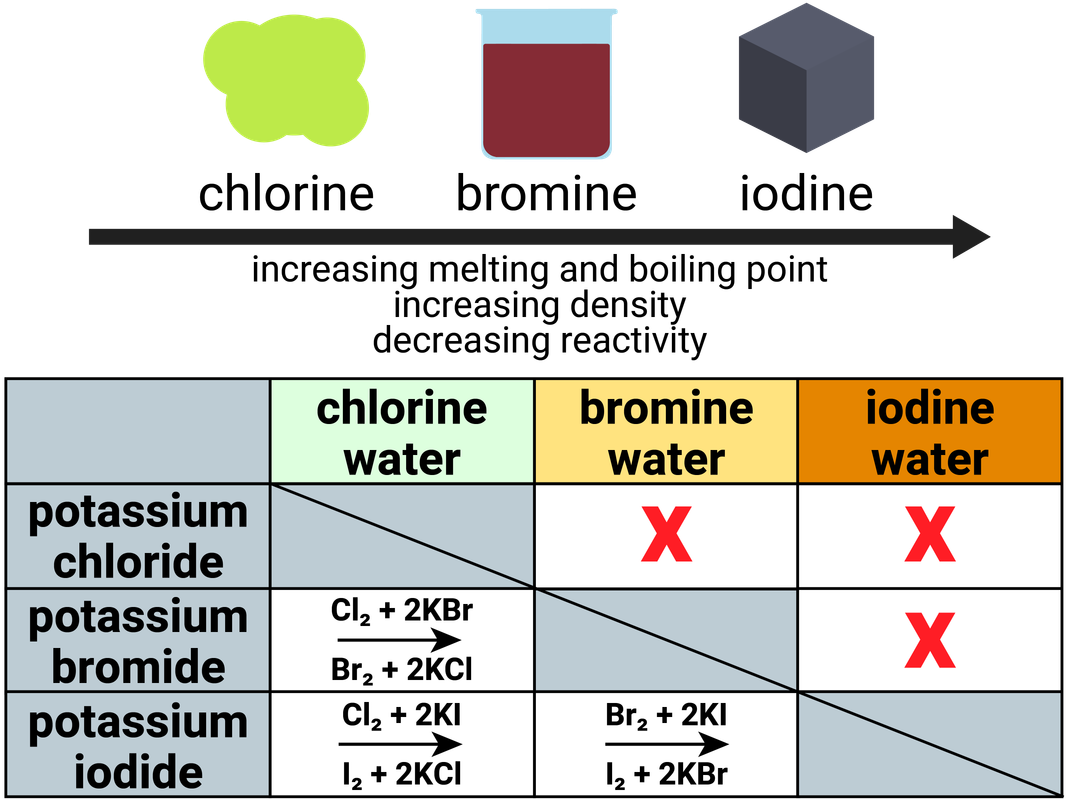
- use a dropping pipette to add a few drops of potassium chloride solution, KCl(aq), to a column of three wells in a spotting tile
- repeat step 1 with two more columns of wells, using potassium bromide solution, KBr(aq), then potassium iodide solution, KI(aq)
- use a dropping pipette to add a few drops of chlorine solution, Cl2(aq), to a row of three wells in the spotting tile
- repeat step 3 with two more rows of wells, using bromine solution, Br2(aq), then iodine solution, I2(aq)
Results and Analysis
Observe and record what happens in each well at step 3 or 4 in a suitable table.
The table above shows some sample results.
Count the number of reactions seen with each halogen solution.
Use the results in the table to deduce an order of reactivity, starting with the most reactive halogen.








