Introducing the Practical
This practical focuses on recording the pH at intervals when calcium hydroxide or calcium oxide reacts with dilute hydrochloric acid.
An initial mass of the solid must be added to a fixed volume of the acid, and the pH recorded each time more of the solid is added to the acid.
The pH can be recorded using a pH meter, or universal indicator paper with a glass rod used to take a pH measurement at each interval.
Risk Asessment
As a general rule, eye protection (goggles) must be worn for all practicals.
| hazard | possible harm | precaution |
|---|---|---|
| hydrochloric acid |
concentrated acid is corrosive and damages skin and clothes |
use dilute hydrochloric acid (only an irritant, wash hands if spillage) |
| calcium hydroxide powder |
skin irritation, serious eye damage, may cause respiratory irritation | wear gloves and avoid making a dust |
This risk assessment is provided as an example only, and you must perform your own risk assessment before doing this experiment.
Apparatus
Each group will need:
100 ml beaker
50 ml measuring cylinder
access to a balance (at least 2 d.p.)
spatula
stirring rod
white tile
universal indicator paper
pH colour chart
dilute hydrochloric acid
calcium hydroxide powder
Experiment Set-up
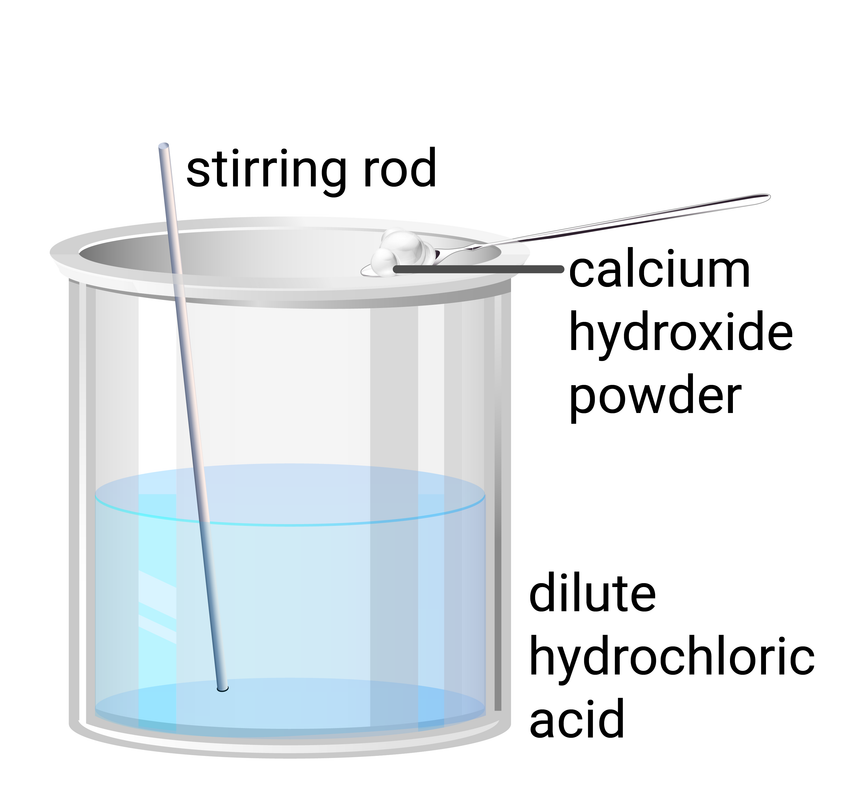
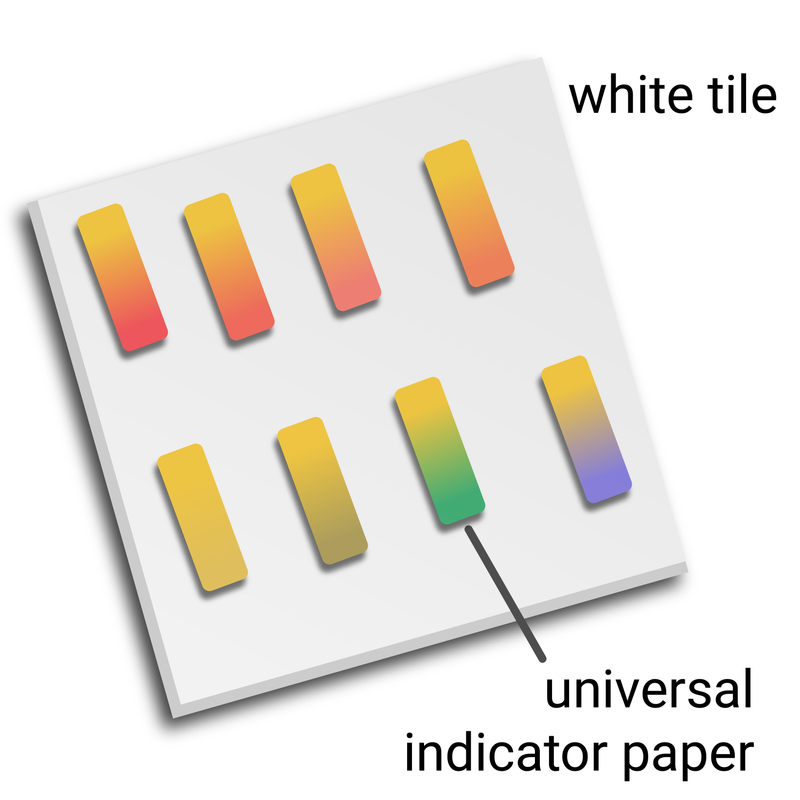
Method
- use a measuring cylinder to add 20 ml of dilute hydrochloric acid to a beaker
- dip a clean glass rod into the contents of the beaker, then use it to transfer a drop of liquid to a piece of universal indicator paper on a white tile
- wait 30 seconds, then match the colour to a pH colour chart - and then record the estimated pH
- add 0.3 g of calcium hydroxide powder to the beaker
- stir thoroughly, then repeat steps 2 and 3
- add another 0.3 g of calcium hydroxide powder to the beaker, and repeat steps 2-4 until you have added a total of 3.0 g of calcium hydroxide powder
Results and Analysis
| mass of calcium hydroxide added (g) | pH of reaction mixture |
|---|---|
| 0.0 | |
| 0.3 | |
| 0.6 | |
| 0.9 | |
| ... |
Plot a graph to show how the change in pH was affected by the amount of calcium hydroxide added.
- pH should be shown on the Y axis (vertical)
- mass of calcium hydroxide should be shown on the X axis (horizontal)
Use the graph to estimate the number of spatulas of calcium hydroxide needed to produce a neutral solution.








