Aims of Experiment
Analysing and purifying a sample of water and making it safe to drink
In this experiment you will:
- analyse a water sample
- purify a water sample by distillation
Risk Asessment
As a general rule, eye protection (goggles) must be worn for all practicals.
| hazard | possible harm | precaution |
|---|---|---|
| acidic/alkaline water samples |
irritating to eyes or skin |
avoid contact, rinse off skin if contact occurs |
| Bunsen burner/hot apparatus |
burns, flame |
allow time to cool before touching, tie back hair/tuck in loose clothes |
This risk assessment is provided as an example only, and you must perform your own risk assessment before doing this experiment.
Apparatus
Each group will need:
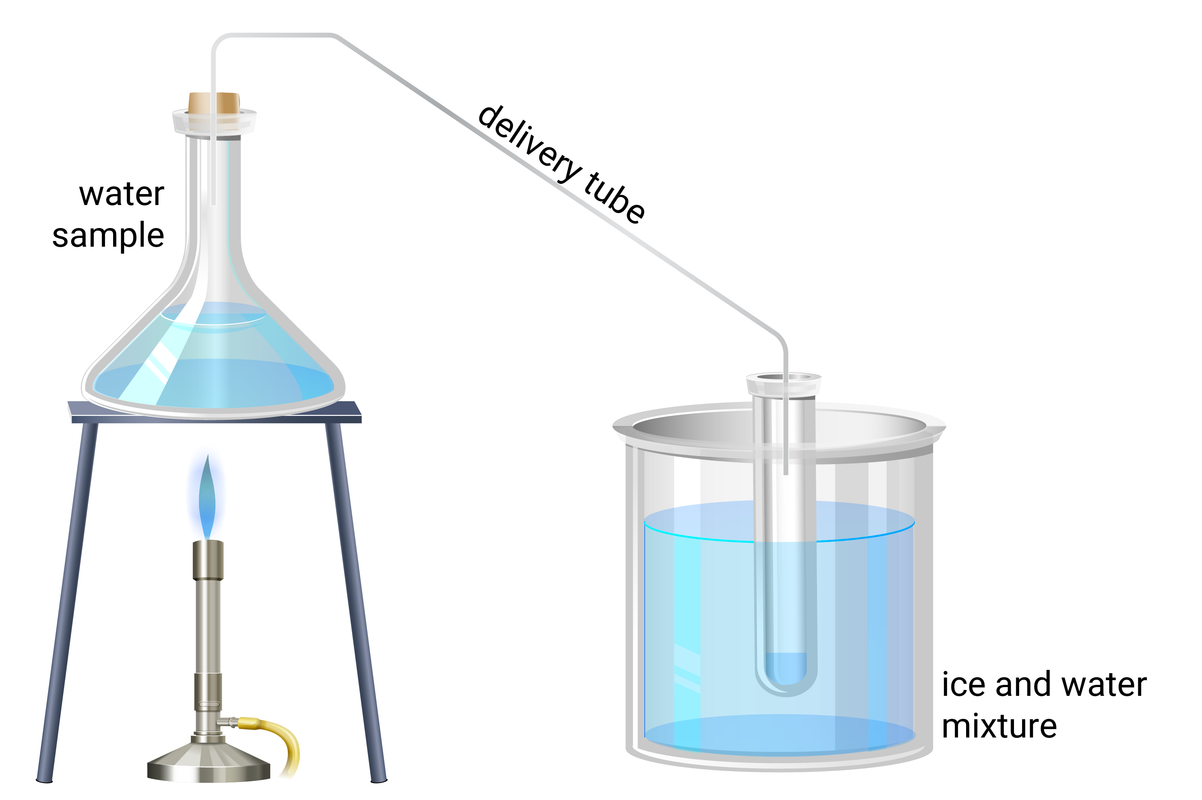
a conical flask with a two‐hole bung with a thermometer and delivery tube
test tube
beaker (250 or 400 ml)
crushed ice
Bunsen burner
tripod
gauze
heat‐resistant mat
3 samples of water
antibumping granules
Experiment Set-up
Part 1

Part 2
Method
Part 1
- use universal indicator paper to measure the pH of the water sample
- weigh an empty evaporating basin (record to two decimal places)
- use a measuring cyclinder to pour 10 ml of water sample 1 into the evaporating basin
- heat the evaporating basin, over a water bath, using a Bunsen burner
- continue heating until the majority of the water has evaporated, then leave on the side until the remaining water has evaporated
- weigh the cooled evaporating basin again and record the mass of the solids that were dissolved in the water
- repeat steps 1-5 for water samples 2 and 3
Part 2
- using a measuring cyclinder pour 10 ml of water sample 1 into a conical flask, and set up the apparatus for distillation (shown above)
- heat the water until it boils, then reduce the heat so it boils gently
- heat the water until about 1 cm depth of water is collected in the test tube
- analyse the water you have distilled by determining its boiling point
- repeat steps 1-4 for water samples 2 and 3
Results and Analysis
Part 1
| pH | mass of dissolved solids (g) | |
|---|---|---|
| sample 1 | ||
| sample 2 | ||
| sample 3 | ||
Part 2
| boiling point (oC) | |
|---|---|
| sample 1 | |
| sample 2 | |
| sample 3 | |
Use the following key words to write a paragraph to explain how you have collected a sample of pure water: evaporation, condensation, gas, liquid, boiling, temperature.
What is the difference between pure water and potable water?
Exam Question and Model Answer
Some countries produce drinking water from sea water. Describe how you can distil salt solution to produce and collect pure water.
How could the water be tested to show it's pure?
[6 marks]
Level 1 (1-2 marks)
Heat the sample of sea water until pure water collects in the test tube, it's boiling point will be at 100°C.
Level 2 (3-4 marks)
Pour a sample of the sea water into a boiling tube, and connect to a delivery tube.
Suspend the delivery tube into a test tube (surrounded by ice and water in a beaker), and set up a Bunsen burner under the boiling tube.
Heat the sample of sea water until pure water collects in the test tube.
Level 3 (5-6 marks)
Pour a sample of the sea water into a boiling tube, and connect to a delivery tube.
Suspend the delivery tube into a test tube (surrounded by ice and water in a beaker), and set up a Bunsen burner under the boiling tube.
Heat the sample of sea water until pure water collects in the test tube.
To test if the water collected is pure, it's boiling point will be at 100°C.








